A research team from Ulsan National Institute of Science and Technology (UNIST) has introduced a groundbreaking technology that can decompose almost all nitrous oxide (N2O) at ambient temperatures. This innovative method employs mechanical impacts and friction to efficiently address nitrous oxide emissions produced by engine exhaust and various chemical processes, representing a significant advancement in efforts to mitigate greenhouse gases and promote carbon neutrality.
The findings, led by Professor Jong-Beom Baek and his team from the School of Energy and Chemical Engineering at UNIST, were published in the journal Advanced Materials. Nitrous oxide, a gas frequently released from chemical manufacturing and vehicle exhaust, has a Global Warming Potential (GWP) that is approximately 310 times greater than that of carbon dioxide, contributing to ozone layer depletion. Traditional thermal catalytic methods have struggled with N2O decomposition, requiring temperatures above 445°C, which leads to high energy consumption.
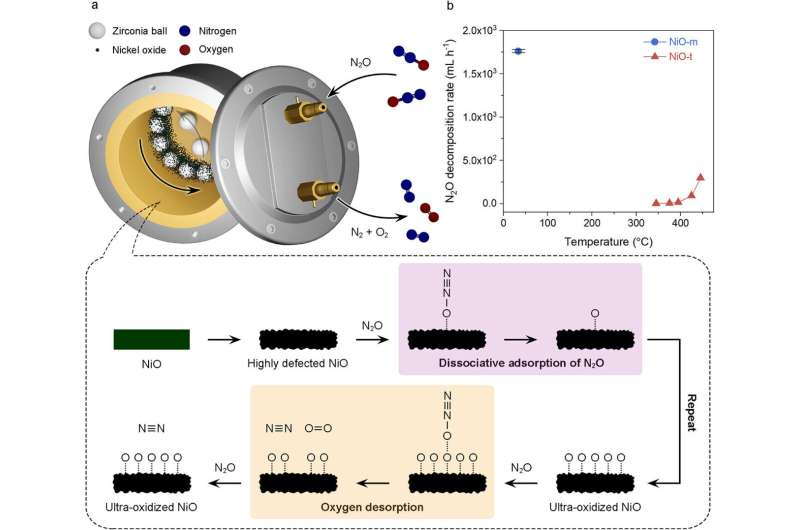
The researchers utilized a ball mill reaction vessel filled with millimeter-sized beads, a nickel oxide (NiO) catalyst, and nitrous oxide gas. By agitating this mixture, the team generated high-energy collisions and friction among the beads, creating dense defects and ultra-oxidized states on the surface of the NiO catalyst. Under these conditions, they achieved rapid decomposition of N2O at low temperatures, a feat that conventional thermal catalysts could not accomplish.
Experimental results revealed that this method can decompose nearly 100% of nitrous oxide at just 42°C, achieving an impressive conversion efficiency of 99.98% and a decomposition rate of 1,761 mL per hour. In comparison, traditional thermocatalytic methods operate at 445°C with a significantly lower conversion rate of 49.16% and an output of 294.9 mL per hour, highlighting a more than sixfold increase in energy efficiency.
The research team further validated the technology”s suitability for real-world applications. In tests simulating emissions from diesel engines, the process removed 95% to 100% of N2O. Additionally, during continuous processing tests designed to replicate large-scale gas treatment facilities, a remarkable conversion rate of approximately 97.6% was sustained. The method also demonstrated stability in the presence of oxygen and moisture, conditions typical of actual exhaust gases.
Economic assessments indicate that this mechanochemical approach is over eight times more cost-effective than existing thermal catalytic processes. Professor Baek remarked, “With the European Union”s upcoming implementation of the Euro 7 emission standards, which include stricter regulations on nitrous oxide, the need for effective removal technologies has become increasingly critical. This innovation addresses N2O emissions effectively from diesel engines, as well as from processes involving nitric and adipic acid production, and ammonia-powered ships, thus supporting efforts toward carbon neutrality and reducing greenhouse gases.”
For further details, refer to the study by Seung-Hyeon Kim et al, “Mechanochemical Nitrous Oxide Decomposition,” published in Advanced Materials in 2025.