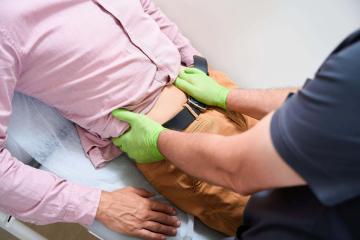
Addenbrooke”s Hospital in Cambridge has become the first medical facility in Europe to offer a groundbreaking treatment for liver cancer, known as histotripsy, outside of clinical trials. This innovative procedure utilizes ultrasound technology to target and destroy cancer cells without surgery, radiation, or chemotherapy, and can be completed in as little as 30 minutes.
The histotripsy treatment was performed on Roger Jackson, an 80-year-old retired sales manager from Bedford, earlier this month. Dr. Teik Choon See, a consultant interventional radiologist at Cambridge University Hospitals NHS Foundation Trust (CUH), led the procedure. Dr. See stated, “Histotripsy represents a major and exciting step forward in cancer treatment.” He emphasized that this technique allows for more precise targeting of tumors while preserving healthy surrounding tissue, providing a safer and quicker alternative to traditional cancer therapies.
One compelling aspect of histotripsy is its potential to activate the patient”s immune response after the ultrasound waves break apart the tumor, which may assist in eliminating any remaining cancerous cells. Mr. Jackson expressed his gratitude, saying, “I feel privileged to be the first NHS patient and to receive this care was an amazing experience.” He noted the impressive nature of using sound waves to treat cancer without the burden of extensive surgery during a stressful time.
This inaugural treatment follows the rapid authorization of histotripsy technology under the Unmet Clinical Need Authorisation (Ucna) in Great Britain, governed by the Medicines and Healthcare Products Regulatory Agency (MHRA). This pathway facilitates access to innovative medical devices under specific conditions prior to receiving full regulatory approval.
Initially, Addenbrooke”s Hospital will provide histotripsy treatment to select patients with primary and secondary liver cancers. Roland Sinker, chief executive of CUH, remarked, “Histotripsy represents a hugely exciting and new era of cancer innovation and care.” He noted that with faster recovery times and shorter hospital stays, the procedure alleviates pressure on hospital resources and allows surgeons to concentrate on more complex cases, thereby reducing waiting times for patients.
Health Secretary Wes Streeting highlighted the significance of this development, stating that it “marks the beginning of a new generation in cancer treatment.” He added, “We are lighting the fuse beneath the technological revolution, transforming care for NHS patients.” Streeting emphasized the government”s efforts to streamline approval processes to ensure this cutting-edge treatment is quickly available to patients, making British patients the first in Europe to benefit from this advancement.
The histotripsy technology, which originated in the United States, has already treated over 2,000 patients globally since receiving Food and Drug Administration approval for the destruction of liver tumors in 2023. Ongoing studies are exploring its effectiveness for various other cancer types.