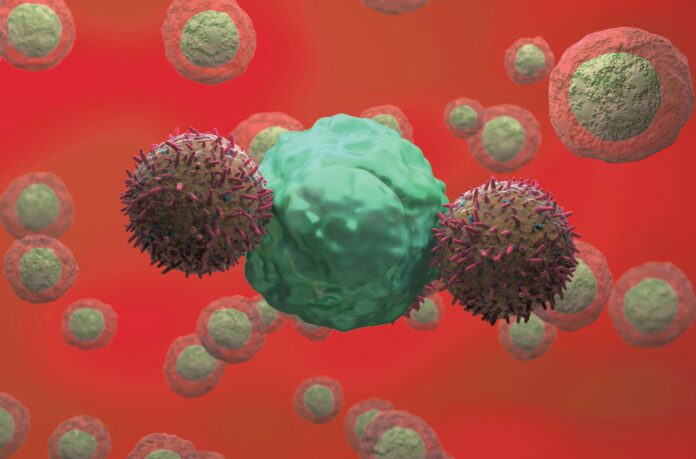
Scientists at École Polytechnique Fédérale Lausanne (EPFL) and UNIL-CHUV, the University Hospital Lausanne, have created a novel computational method for designing synthetic receptors that enhance the ability of engineered T cells to respond to tumors. Led by Dr. Patrick Barth at EPFL and Dr. Caroline Arber at UNIL-CHUV, the research team developed a platform to construct synthetic protein receptors from the ground up.
These custom receptors, termed T-SenSERs (tumor microenvironment-sensing switch receptors), are engineered to recognize soluble signals prevalent in tumors. They convert these signals into co-stimulatory or cytokine-like signals, which significantly enhance T cell activity against cancer cells. The findings have been published in Nature Biomedical Engineering, in a paper titled “Computational design of synthetic receptors with programmable signalling activity for enhanced cancer T cell therapy.” The researchers concluded that their work lays a foundation for the rapid development of synthetic biosensors tailored for various cell engineering applications.
Engineered T cell therapies, particularly those utilizing chimeric antigen receptors (CAR T cells), have shown considerable promise in treating blood-related cancers. These bioengineered T cells represent a breakthrough in cancer treatment. However, their impact has not been as pronounced in solid tumors, such as those found in the breast, lung, and prostate.
For sustained anti-tumor responses, engineered CAR- or T-cell receptor (TCR)-transgenic T cells must identify and eliminate tumor cells while also receiving specific co-stimulatory and cytokine signals from their surroundings. A significant challenge arises from the tumor microenvironment (TME), which consists of a mix of cells and molecules that can suppress immune responses. As noted by the researchers, “The tumor microenvironment plays a key role in tumor progression, and soluble and cellular TME components can limit CAR T cell function and persistence.”
In many solid tumors, inhibitory signals are prevalent, while supportive signals that encourage T cell activity are either weak or non-existent. Since engineered T cells depend on these environmental cues to maintain their function, they often underperform. This has prompted scientists to investigate ways to integrate additional receptors into T cells, enabling them to detect tumor-specific signals and respond more robustly.
The challenge lies in the complexity of constructing custom signaling proteins. Current methods predominantly rely on trial-and-error, making it difficult to predict how these synthetic receptors will behave in tumor environments. “Targeting soluble TME factors to enhance anti-tumor responses of engineered T cells through chimeric receptors is not broadly explored owing to the unpredictable signaling characteristics of synthetic protein receptors,” the authors noted.
Barth, Arber, and their team have now advanced a computational platform that assembles artificial receptors by designing and combining various protein domains, akin to building with molecular Legos. Each receptor comprises an external domain that binds tumor-associated signals, a middle region that transmits this signal across the cell membrane, and an internal domain that activates beneficial functions within the T cell. “We developed a computational approach for the bottom-up assembly and design of multi-domain receptors with programmable input–output signaling functions,” they stated.
Using this innovative platform, the team created two families of T-SenSERs: one that responds to VEGF, a protein promoting blood vessel growth commonly found in tumors, and another that reacts to CSF1, a protein that adversely affects immune cell behavior in tumors. After designing 18 variations, they selected the most effective versions based on simulations and laboratory tests.
In experimental settings, T cells equipped with both a CAR and a T-SenSER exhibited a stronger response to tumors compared to CAR T cells alone, demonstrating ligand-specific activities consistent with the signaling programs developed through their design method. The VEGF-sensing receptor (designated VMR) activated the T cell solely in the presence of VEGF, while the CSF1-sensing receptor (CMR) provided a minor baseline boost even without CSF1 but significantly intensified its effect when the ligand was present.
In mouse models of lung cancer and myeloma, T cells with these synthetic receptors showed improved tumor control and extended survival rates. “The combination of CAR and T-SenSER in human T cells enhances anti-tumor responses in models of lung cancer and multiple myeloma, in a VEGF- or CSF1-dependent manner,” the researchers noted.
Crucially, the team discovered that their design approach allowed for fine-tuning of the receptor behaviors, enabling choices between always-on, ligand-dependent, or intermediate functions. “Based on rational design principles of signal transduction, our technology can engineer synthetic chimeric receptor structures with predictable and desired signaling output, setting the stage for the broader and more efficient development of biosensors with novel input–output functions,” they stated.
Barth remarked that this research represents the first successful demonstration of computationally designed single-pass, multi-domain receptors with programmable signaling functions. It paves the way for accelerated development of synthetic biosensors equipped with tailored sensing and response capabilities, applicable to both basic and translational cell engineering.
The authors highlighted the broader potential applications of their T-SenSER technology, envisioning future uses in various therapeutic T cell products such as CAR T cells with different endo-domains, TCR-T cells, tumor-infiltrating lymphocytes, or virus-specific T cells, where significant improvements could be achieved through TME-specific enhancements of the anti-tumor response.