A research team at Beihang University has innovated a ferromanganese oxide-modified biochar that demonstrates exceptional capability in removing copper-citrate (CuCA) complexes from polluted water. This novel material, featuring nanoparticles of Mn3O4 and (FeO)0.099(MnO)0.901 measuring around 100 nanometers, achieved an impressive copper removal rate of 99.5% along with a total organic carbon reduction of 92.6%.
The adsorption process is characterized by a combination of chemical bonding facilitated by oxygen-containing functional groups and physical adsorption via the biochar”s porous structure. As freshwater scarcity increases globally, the demand for effective wastewater recycling and treatment solutions has become critical. Traditional treatment methods primarily target free metal ions and often neglect stable metal complexes prevalent in industrial and municipal discharges. These complexes, frequently associated with citric acid or other organic compounds, are resistant to degradation and can pose long-term environmental and health hazards.
Copper-citrate complexes are particularly significant in sectors like electroplating, textile dyeing, and various household applications. Conventional methods, such as precipitation and ion exchange, are frequently inadequate for efficiently removing these stable complexes. Although biochar is recognized as a cost-effective and environmentally friendly adsorbent, its effectiveness has been limited by a lack of active sites and selective adsorption capabilities. Enhancements in biochar design are necessary to address these limitations.
The study, published in Biochar X on October 14, 2025, by a team led by Wenhong Fan, details the synthesis of ferromanganese oxide-modified biochar (FMBC-600) using impregnation and high-temperature calcination techniques. The research systematically evaluated the material”s structure and its efficacy in extracting CuCA complexes from aqueous solutions. Field emission scanning electron microscopy (FE-SEM) showed that the modification process transformed the originally smooth biochar surface into a rough texture uniformly coated with nanoparticles. Energy-dispersive X-ray spectroscopy (EDS) confirmed the incorporation of iron and manganese, along with the deposition of copper after adsorption.
Fourier-transform infrared spectroscopy (FTIR) and X-ray diffraction (XRD) analyses revealed the presence of various functional groups, including hydroxyl and aromatic groups, alongside crystalline phases of Mn3O4 and (FeO)0.099(MnO)0.901. X-ray photoelectron spectroscopy (XPS) indicated that the adsorption process involves redox interactions between iron and manganese, facilitating electron transfer and the formation of surface complexes.
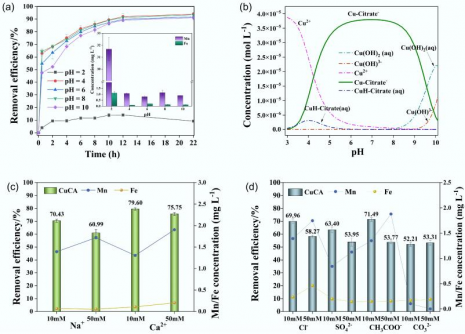
Moreover, nitrogen adsorption-desorption studies showed an increase in pore volume, enhancing the material”s adsorption capacity. Optimal conditions for copper removal were identified as an iron-to-manganese ratio of 1:4, manganese concentration of 0.03 M, and pyrolysis temperature of 600 °C. Under these conditions, the biochar achieved a copper removal efficiency of 99.5% and a total organic carbon removal of 92.6%. The adsorption process was rapid, reaching completion within 30 minutes, and remained stable across a pH range of 4 to 10. FMBC-600 also demonstrated resilience against interference from competing ions such as sodium, calcium, chloride, and sulfate, maintaining high selectivity and performance.
Kinetic modeling indicates that the adsorption process follows a pseudo-second-order equation, suggesting that chemisorption is the main mechanism at play. The Freundlich isotherm model describes the heterogeneous multilayer adsorption, which is enhanced at elevated temperatures. Regeneration tests showed that the material retained about 80% of its efficiency after two cycles of use.
Overall, the uniformly structured FMBC-600 exhibits excellent stability, reusability, and high adsorption efficiency, positioning it as a scalable and affordable solution for the removal of persistent heavy metal complexes from wastewater. This advancement signifies a substantial step forward in sustainable water treatment technologies. The straightforward and cost-effective production process of this modified biochar makes it particularly suitable for large-scale applications in managing industrial effluents, particularly within the electroplating, dyeing, and chemical manufacturing sectors. Furthermore, this technology has potential applications in soil remediation, contributing to the reduction of heavy metal accumulation in agricultural lands.
Compared to traditional adsorbents, FMBC-600 provides enhanced selectivity, stability, and reusability. Its ability to function effectively under diverse pH and ionic conditions makes it practical for real-world applications where water chemistry can be complex. Integrating this innovative biochar into wastewater treatment systems could significantly mitigate metal contamination, thus aiding in the pursuit of global clean water and sustainability objectives.