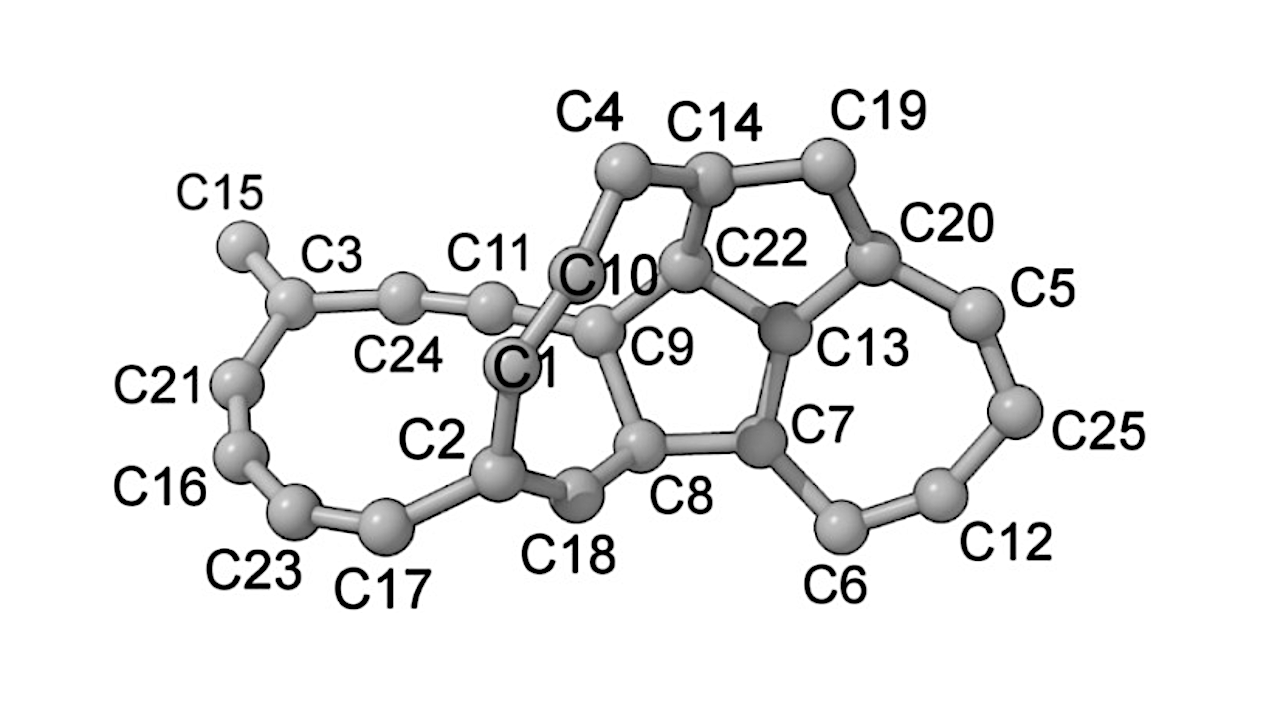
In a significant advancement in chemistry, researchers have successfully developed a computational method that accurately predicts the optimal ligand for a photochemical palladium catalyst. This breakthrough opens the door to new radical reactions involving alkyl ketones, which are prevalent components in a wide variety of organic molecules.
Chemists have long sought innovative techniques to utilize ketones effectively in the formation of chemical bonds. One of the most challenging reactions associated with ketones is the one-electron reduction, a process that results in the production of ketyl radicals. These radicals are crucial intermediates in various organic reactions, making the ability to predict suitable ligands for catalysis particularly valuable.
The new computational approach not only enhances the efficiency of these reactions but also improves the overall understanding of how ligands can influence catalytic activity. This development is poised to aid chemists in designing more effective catalysts for a range of applications, thereby advancing research in organic synthesis.
Overall, this innovative method represents a pivotal step forward in the field of chemistry, providing valuable insights that could transform how researchers approach the utilization of ketones in chemical processes.