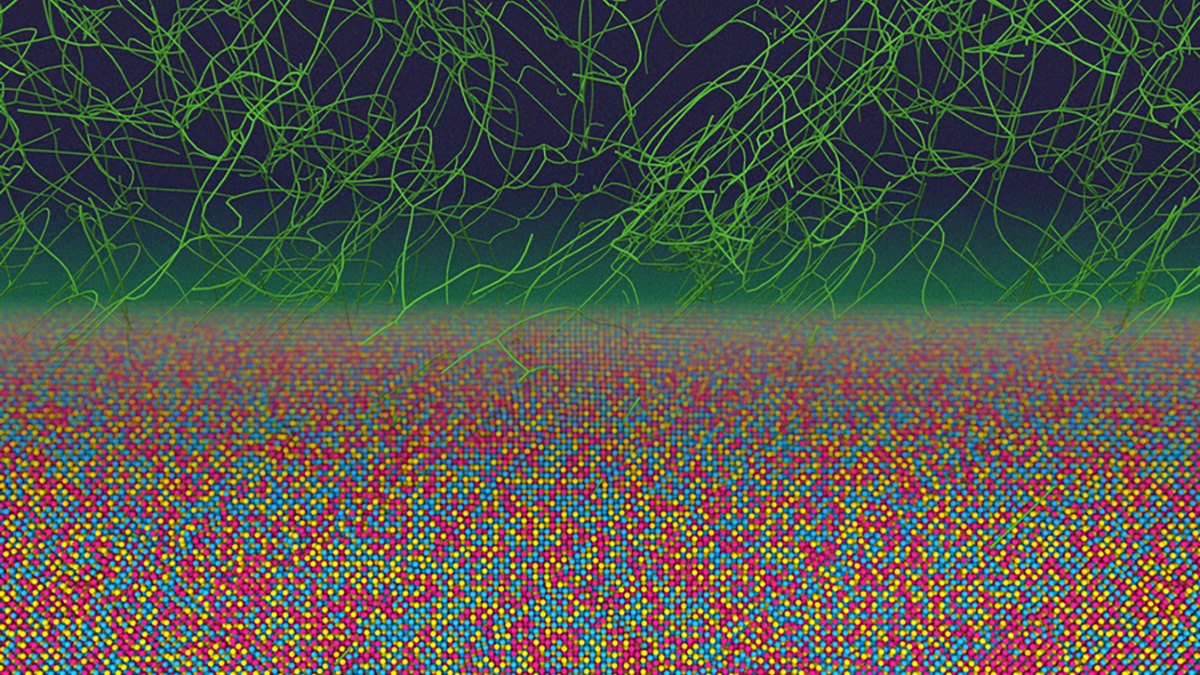
Recent research from the Massachusetts Institute of Technology has revealed surprising atomic patterns within metal alloys that challenge conventional manufacturing wisdom. Traditionally, it was believed that the atoms in metal alloys were arranged randomly, but this new study demonstrates that certain atomic configurations can persist even after extensive processing.
The findings, published in the journal Nature Communications, indicate that these hidden patterns could be crucial for enhancing the properties of metals, such as mechanical strength, durability, and resistance to radiation. In lab experiments, researchers employed advanced computer simulations to analyze the behavior of millions of atoms in an alloy composed of chromium, cobalt, and nickel (CrCoNi) during common manufacturing processes, including rapid cooling and stretching.
According to materials scientist Rodrigo Freitas, “This is the first paper showing these non-equilibrium states that are retained in the metal. Right now, this chemical order is not something we”re controlling for or paying attention to when we manufacture metals.” The study focused on what researchers refer to as chemical short-range order (SRO), which describes the specific arrangements that atoms adopt in metal alloys.
The researchers identified familiar atomic patterns that surprisingly persisted following rapid deformations, alongside new configurations dubbed “far-from-equilibrium states.” These states are maintained by defects or dislocations that occur in the crystal structure of metals during heating, cooling, or stretching processes. Freitas remarked, “These defects have chemical preferences that guide how they move. They look for low-energy pathways, so given a choice between breaking chemical bonds, they tend to break the weakest bonds, and it”s not completely random.”
This discovery has significant implications for the manufacturing of metals, suggesting that the inherent atomic order cannot be entirely randomized regardless of processing methods. Freitas noted, “The conclusion is: you can never completely randomize the atoms in a metal. It doesn”t matter how you process it.” The unexpected persistence of these atomic patterns opens new avenues for fine-tuning the properties of metal alloys, with potential applications ranging from nuclear reactors to aerospace technologies.
As researchers continue to explore these findings, there is optimism that understanding these atomic arrangements will lead to innovative ways to optimize metal performance in various high-tech applications.