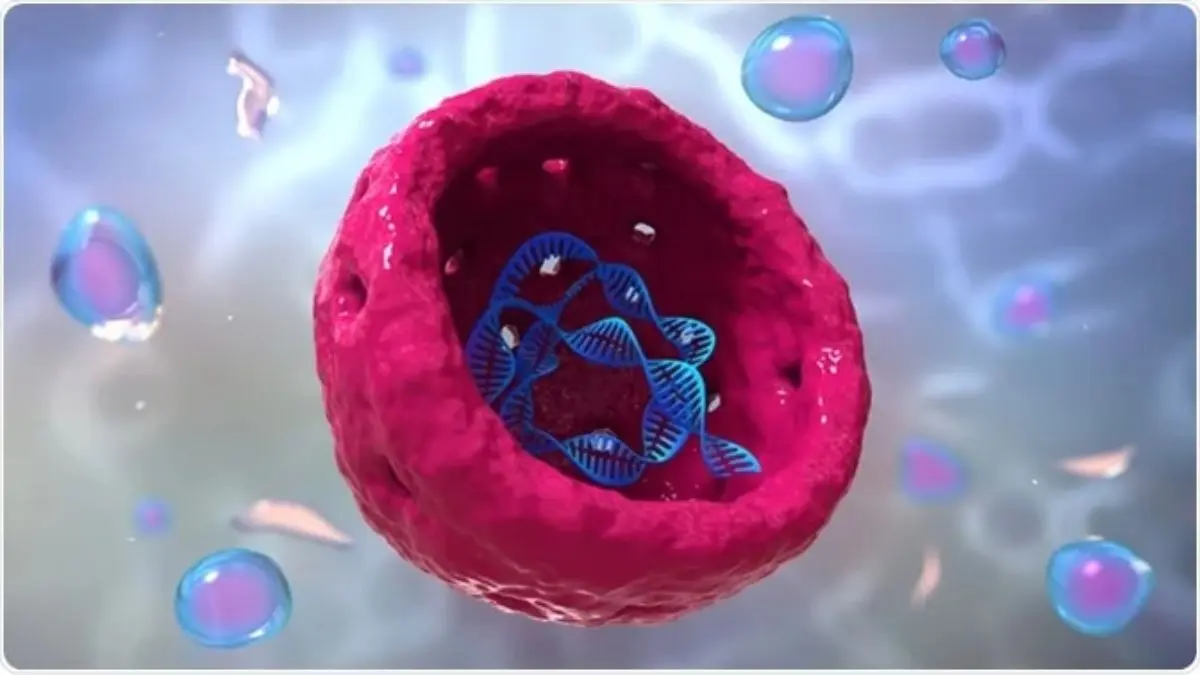
Israeli and American researchers have made significant strides in understanding how tiny gateways within human cells manage the flow of substances into and out of the cell nucleus. This breakthrough could enhance our understanding of various diseases, including cancer, Alzheimer”s, and amyotrophic lateral sclerosis (ALS). The findings were announced by the Hebrew University of Jerusalem.
A collaborative effort involving scientists from the Hebrew University, the Quantitative Biosciences Institute at the University of California, San Francisco, The Rockefeller University, and the Albert Einstein College of Medicine has revealed that these gateways, known as nuclear pore complexes (NPCs), utilize a dynamic protein framework along with specialized molecular “passports” to facilitate swift and precise molecular transport.
Dr. Barak Raveh, the lead author of the study, described the model developed by the team as functioning like a “virtual microscope,” allowing researchers to observe the operations of these tiny structures, which measure approximately one five-hundredth the width of a human hair. “By integrating numerous independent experiments and conducting computer simulations, we can finally visualize how this gate functions second by second,” he explained.
Raveh likened NPCs to advanced security checkpoints that can allow millions of molecules to pass per minute while maintaining a high level of selectivity. For years, scientists struggled to comprehend how NPCs managed to exhibit both speed and selectivity due to their minuscule size, which makes direct observation challenging.
Previous theories depicted these gateways as rigid barriers or sponge-like filters, neither of which could adequately clarify how NPCs permit large molecular structures to cross while blocking smaller entities. The newly proposed model combines experimental insights and computational methods to reveal the processes occurring at the molecular level within milliseconds.
Inside the NPCs exists a dense, constantly shifting “forest” of protein chains known as FG repeats. These chains create a crowded atmosphere that naturally obstructs unescorted molecules, while allowing smaller entities to pass through. Larger molecules can traverse the NPCs if they are escorted by nuclear transport receptors, which serve as molecular “passports” that briefly interact with the FG chains to guide their cargo.
Professor Michael Rout from The Rockefeller University noted, “The FG repeat chains are in continuous motion, creating a dynamic and congested environment. The transport resembles a constantly evolving dance across a bridge. Only those carrying the proper partners— the receptors— can proceed through. Without them, others are turned away.”
This model addresses a longstanding question about how NPCs can permit substantial molecular complexes to pass while excluding smaller ones. Professor Andrej Sali from the Quantitative Biosciences Institute remarked, “Our model offers the first clear explanation of how NPCs achieve this extraordinary selectivity, paving the way for new medical and biotechnological advancements.”
According to Professor David Cowburn of the Albert Einstein College of Medicine, the discoveries have immediate relevance for understanding diseases linked to dysfunctional nuclear transport systems, such as ALS, Alzheimer”s, and various cancers. Moreover, this research may lead to practical applications, including the design of drugs that regulate molecular transport within cells or the creation of synthetic nanopores that imitate NPCs, potentially allowing for targeted treatments to be delivered directly to the nucleus.
The model not only accurately predicted previously unobserved transport behaviors but also demonstrated that transient interactions between receptors and FG chains contribute to the system”s efficiency. Its inherent redundancy ensures that NPCs remain dependable even under stress, which helps explain the evolutionary success of this system. The findings are published in the peer-reviewed journal Proceedings of the National Academy of Sciences.