Researchers at the University of California Berkeley”s Innovative Genomics Institute have developed a novel method to enhance the nuclear entry of the CRISPR-Cas9 protein, aiming to improve its efficacy in gene editing. This advancement could significantly impact the development of gene-edited cell therapies, potentially lowering manufacturing costs and accelerating treatment availability for patients.
Gene editing tools must be both precise and efficient, and CRISPR-Cas9 has emerged as a leading choice in recent years. Unlike traditional viral vectors that randomly integrate DNA into the genome, CRISPR-Cas9 allows for targeted modifications by cutting DNA at specific locations. However, its effectiveness in clinical applications has been hindered by the challenge of ensuring sufficient Cas9 protein reaches the cell nucleus, where DNA modification occurs.
Current strategies typically involve attaching nuclear localization signal (NLS) motifs to the ends of Cas9 to aid its transport into the nucleus. Unfortunately, this method has proven inefficient, as a significant portion of the Cas9 introduced into cells fails to enter the nucleus. Improving this aspect is crucial for enhancing therapeutic outcomes, where even small improvements can lead to more effective treatments.
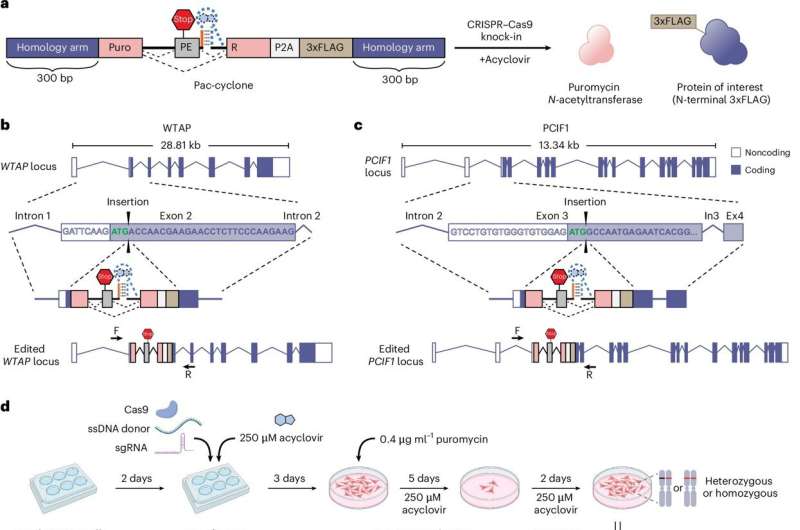
The Berkeley team proposed an innovative solution by incorporating additional NLS motifs into internal loops of the Cas9 protein, rather than simply extending the terminal ends. Their research revealed that this approach not only increased the distribution of NLS motifs across the protein but also maintained its stability and functionality. Each hairpin internal NLS (hiNLS) module consists of two NLS motifs arranged in tandem, increasing the probability that Cas9 remains bound during its journey into the nucleus.
To assess the effectiveness of their hiNLS-Cas9 variants, the researchers conducted experiments on primary human T cells, which are integral to various cell therapies, including chimeric antigen receptor (CAR) T cells. They used gRNA-Cas9 ribonucleoprotein complexes (RNPs) targeting genes relevant to clinical applications, such as beta-2 microglobulin (b2M) and T-cell receptor alpha chain (TRAC). Disrupting b2M is known to enhance the immune evasion of T cells, while targeting TRAC can help prevent adverse graft-versus-host reactions.
The team utilized two delivery methods: traditional electroporation and a peptide-mediated delivery approach known as peptide-enabled ribonucleoprotein delivery for CRISPR engineering (PERC). The PERC method is less invasive, improving cell viability while simplifying the delivery process. This is particularly beneficial in clinical settings where maintaining cell health during the editing process is essential.
Results indicated that Cas9 variants with multiple hiNLS modules exhibited significantly improved editing efficiencies. For example, a variant featuring two NLS modules achieved over 80% knockout of the b2M gene in T cells, whereas traditional Cas9 reached approximately 66%. The PERC method also demonstrated competitive knockout rates without compromising T-cell viability.
This study highlights several key advantages of the hiNLS-Cas9 approach for clinical applications. By overcoming the nuclear entry barrier, this method allows for higher editing efficiencies, which could also benefit challenging cell types. Additionally, the hiNLS variants maintained comparable production yields to unmodified Cas9, facilitating scalability for clinical use.
Future developments may involve combining the hiNLS-Cas9 strategy with other advancements, such as virus-like particles or lipid nanoparticles for direct in vivo editing. The researchers also noted some increase in off-target activity with the hiNLS-Cas9, suggesting that integrating high-fidelity Cas9 mutations could optimize both accuracy and efficiency in gene editing.
In conclusion, by focusing on enhancing nuclear localization, researchers have developed a more effective tool that has the potential to revolutionize gene-edited cell therapies, ultimately benefiting developers and patients alike.