Researchers at Weill Cornell Medicine have introduced an innovative and non-toxic technology that enables the regulation of any gene within a cell. This “gene-switch” mechanism empowers scientists to effectively activate or deactivate specific genes, facilitating the exploration of their functions, disease modeling, and the creation of new therapeutic strategies.
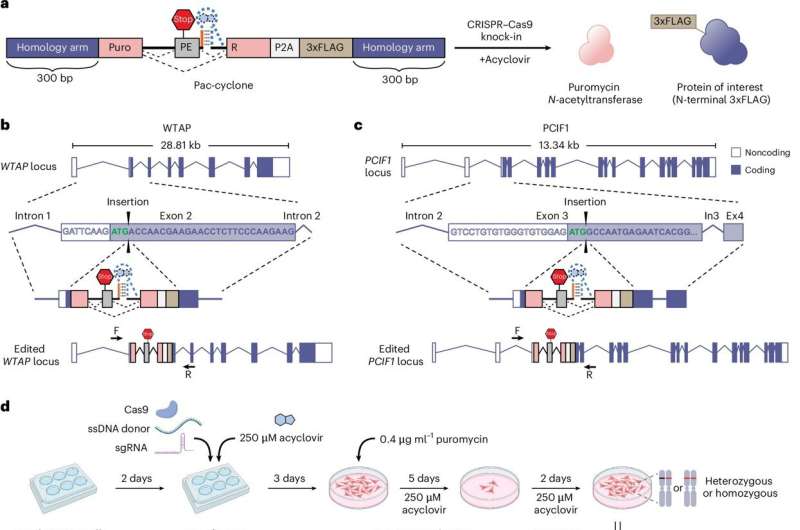
The newly developed tool, known as Cyclone (acyclovir-controlled poison exon), is detailed in a paper published in Nature Methods. This technology draws inspiration from a natural aspect of certain genes, specifically a DNA segment termed a “poison exon,” which can inhibit the translation of a gene into protein under specific circumstances.
To construct Cyclone, the research team engineered a poison exon that can be integrated into any target gene to suppress its expression. When researchers wish to enhance gene activity, they can introduce a drug that binds to the poison exon, allowing activity to resume. Unlike many existing gene-switch tools, Cyclone utilizes the antiviral medication acyclovir, which is considered relatively safe, to activate the target gene.
“We believe the Cyclone concept holds significant promise for various applications that require the safe and precise control of gene activity,” stated Dr. Samie Jaffrey, the Greenberg-Starr Professor in the Department of Pharmacology and a member of the Sandra and Edward Meyer Cancer Center at Weill Cornell Medicine. The first author of the study was Qian Hou, a Ph.D. candidate in the Jaffrey Laboratory.
Cyclone represents a notable advancement compared to existing gene-switch technologies, many of which have considerable limitations. For example, some compounds used to regulate gene expression, such as tetracycline, can produce unwanted, even toxic, effects on cells. Furthermore, traditional tools frequently alter RNA transcripts—the templates used for protein synthesis. In contrast, acyclovir is regarded as safe even at elevated doses and does not modify RNA transcripts or the proteins they encode when activating a target gene.
The researchers demonstrated that Cyclone can increase gene activity from nearly 0% to over 300%, depending on the dosage of acyclovir. They also established that the Cyclone system could function with both artificial genes and naturally occurring genes in cells, and could potentially be compatible with switch molecules beyond acyclovir. This opens up the possibility of employing different Cyclone-type systems to manipulate multiple genes at once.
The team notes that future applications of Cyclone-type systems could include gene therapies, serving as a reversible safety mechanism to regulate the activity of therapeutic genes.
More information can be found in the study authored by Qian Hou et al, titled “A portable poison exon for small-molecule control of mammalian gene expression,” published in Nature Methods in 2025.