Have you ever wondered why water does not seep out of a glass even though the atoms in the glass are theoretically spaced apart? This intriguing question arises from a misconception that the atoms in the glass are empty while those in the water are not. In reality, all atoms share fundamental characteristics, regardless of whether they belong to a solid like glass or a liquid like water.
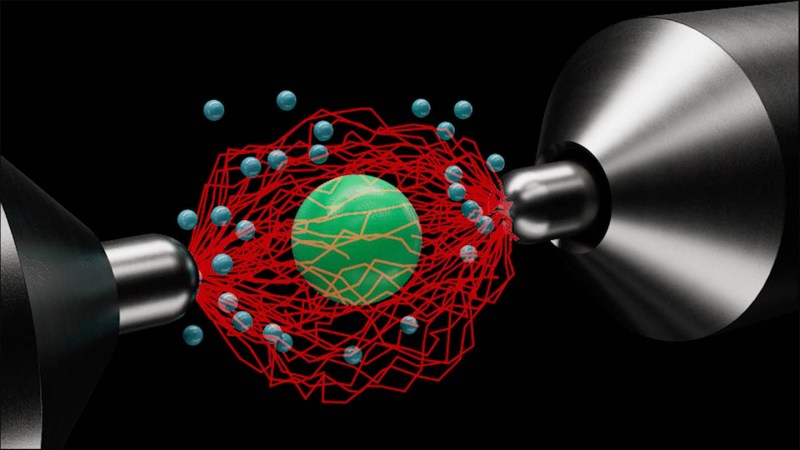
Crucially, atoms are not truly empty. Surrounding each atom”s nucleus is a probability cloud of one or more electrons. These electrons generate a quantum electromagnetic field that acts as a barrier between atoms, including those in water. Therefore, the reason water does not escape from a glass is closely tied to the interactions between these atomic electrons.
When considering why water remains contained, it is essential to recognize the role of electron repulsion and attraction. Electrons, which all carry a negative charge, repel one another. Conversely, they are attracted to positively charged particles. This dynamic means that water molecules are drawn to the glass while also remaining cohesive with one another due to these electromagnetic forces.
This situation has an analogy in adolescence, where young people feel a strong attachment to their families while also forming new bonds outside of their homes. In atomic terms, when an electron from a water molecule attempts to occupy a space already filled by an electron in a glass atom, it cannot do so due to the Pauli exclusion principle. This principle states that no two electrons can occupy the same quantum state simultaneously, effectively preventing water atoms from penetrating the glass structure.
Ruth Lazkoz, a physics professor at the University of the Basque Country, emphasizes that the presence of these electrons creates barriers in all types of matter. As a result, the atomic structure of both the glass and the water plays a significant role in maintaining the integrity of the water within the glass.
This scientific inquiry is part of a weekly column, “Scientists Answer,” which addresses reader questions about science and technology. Sponsored by the L”Oréal-Unesco “For Women in Science” program and Bristol Myers Squibb, the initiative features women scientists answering queries submitted by the public.
For further questions, readers can reach out via email at [email protected].